- Blog
- Xnxx gay spy xams
- Fat black gay porn sites
- Where can gay men donate blood
- Electric six gay bar costume
- Best gay chat app japan
- Cruise gay meme funny
- So yeah your gay meme
- Gay anime manga hentai
- Funny gay pride shirt
- He has big gay meme
- Hand jobs gay cum shot comp
- Rudy gay spurs dunk
- Gay snapchat trade groups

We take no position on whether the FDA’s blood donation policy is discriminatory against gay and bisexual men.
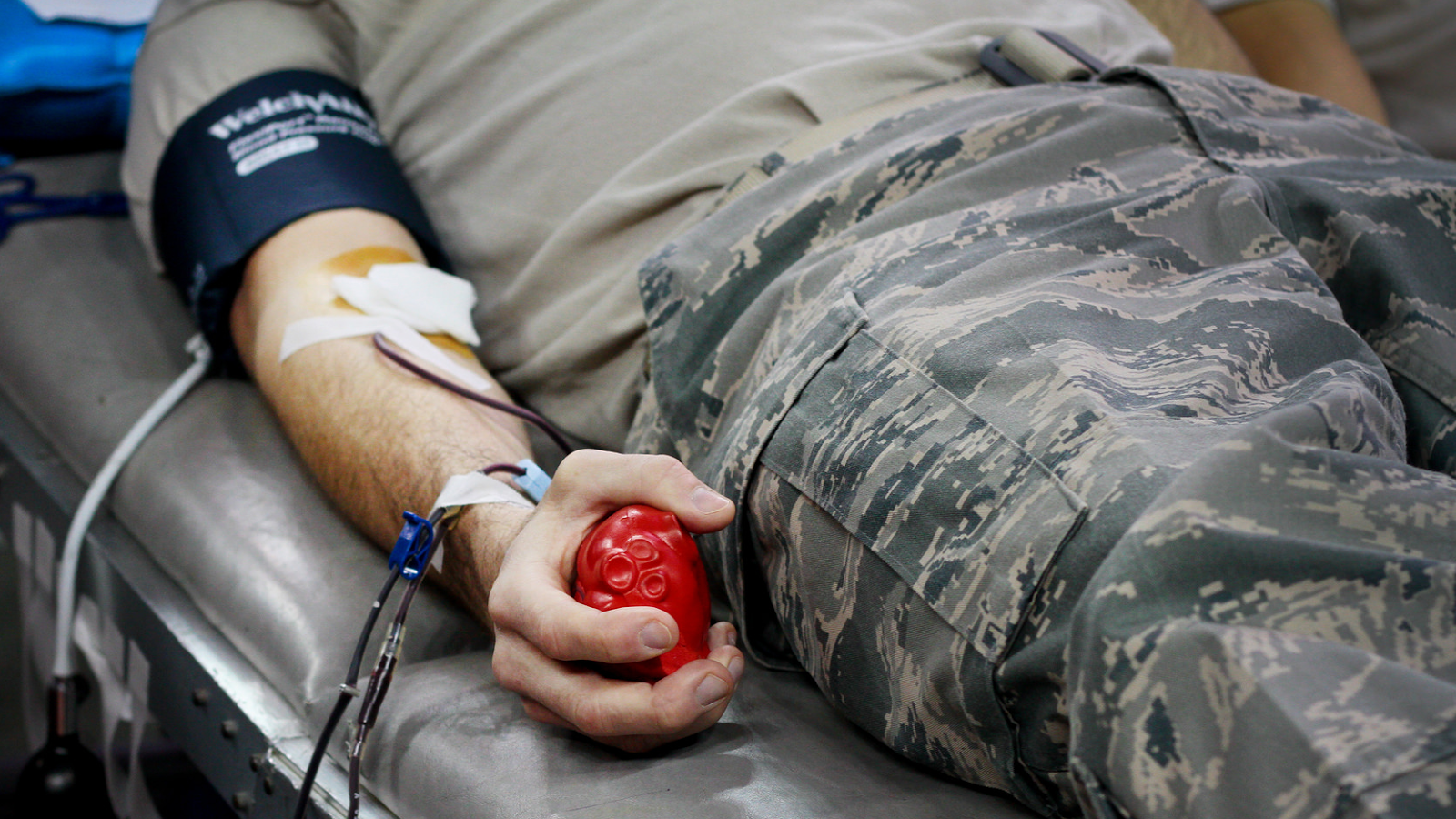
How much scientific evidence is enough for policy change is matter of opinion. Still, the FDA argues there is “inadequate data” to support an individual risk-based assessment for MSM blood donations for the U.S. They also cite the FDA’s own revised policy document, which states that HIV prevalence in MSM blood donors is much lower than HIV prevalence in the MSM population as a whole. Before this change, Australian state and territory policies ranged from a lifetime ban to a five-year deferral for MSM.īut the House members and other critics of the FDA policy have argued that a one-year deferral for MSM is unnecessary because advances in HIV testing have substantially reduced the risk of transmitting the virus via blood transfusions.

For example, between 19, Australia changed its policy to a one-year deferral for MSM and saw no increase in HIV transmissions via blood transfusions. The FDA also uses evidence from other countries to support its case. While all blood donations are tested for HIV, current tests don’t work until approximately nine days after the virus is first transmitted, a so-called “window period.” Blood donations from MSM during this time pose a risk to the blood supply, the FDA says. The FDA cites the high prevalence of human immunodeficiency virus, or HIV, in the MSM population as support for its one-year deferral policy. The revised policy also defers blood donations from a woman who has had sex with an MSM for one year. 21, 2015, the FDA switched its original 1985 MSM blood donation policy from a ban on any man who has had sex with another man since 1977 to a one-year deferral. However, Lee and others’ proposed alternative – an individual risk-based assessment which would evaluate whether potential donors’ behaviors put them at risk for transfusion-transmissible infections – also has scientific evidence to back it up. In fact, the FDA’s policy is supported by scientific evidence. Barbara Lee said on June 14, for example, that the FDA’s one-year deferral policy, or celibacy, for men who have sex with men (MSM) is “based in fear and stigma, not science.” The White House, on the other hand, claimed on the same day that the FDA’s current policy does “rely on scientific advice.” Days after the mass shooting at a gay nightclub in Orlando, House members and the White House made seemingly contradictory claims concerning the Federal Drug Administration’s blood donation policy for gay and bisexual men.Ĭalifornia Rep.
- Blog
- Xnxx gay spy xams
- Fat black gay porn sites
- Where can gay men donate blood
- Electric six gay bar costume
- Best gay chat app japan
- Cruise gay meme funny
- So yeah your gay meme
- Gay anime manga hentai
- Funny gay pride shirt
- He has big gay meme
- Hand jobs gay cum shot comp
- Rudy gay spurs dunk
- Gay snapchat trade groups
